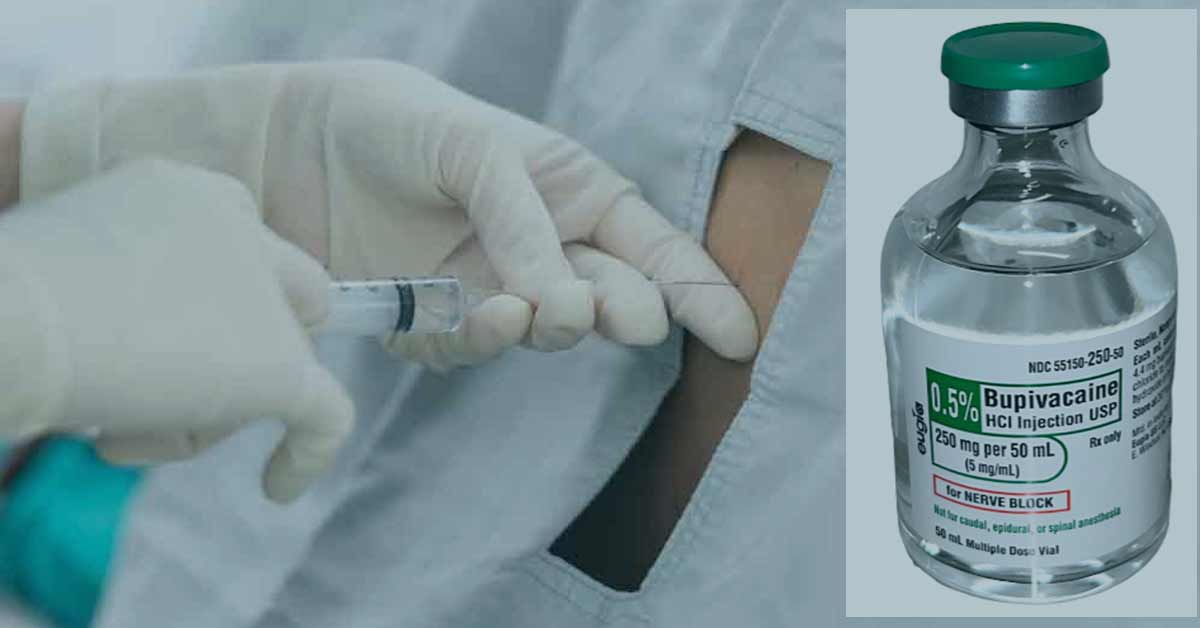
Bupivacaine plain (Marcain, Marcaine) is a long-acting amide-type local anaesthetic drug used for all all types of block except intravenous regional block and obstetric paracervical block. Bupivacaine, first discovered in 1957, is related to mepivacaine.
Local anesthetics generally block the generation of the action potential in nerve cells by increasing the threshold for electrical excitation. They are used in regional anesthesia, epidural anesthesia, spinal anesthesia, and local infiltration.
Bupivacaine plain is a sterile, aqueous, clear, colourless, particle free and isobaric solution for injection. The relative density is 1.004 at 20°C, while the pH is adjusted to to 4.0 to 6.5.
Marcaine plain is slightly hyperbaric (compared to cerebrospinal fluid) at 20°C and slightly hypobaric at 37°C. It is considered to be isobaric as its spread is marginally affected by gravity. It produces lower level of block, for a longer duration, than the hyperbaric solution.
Bupivacaine plain is available in 0.25% (2.5mg/ml), 0.5% (5mg/ml), and 0.75% (7.5mg/ml) concentrations.
Composition of Bupivacaine Plain:
Bupivacaine plain contain the main active ingredient, Bupivacaine hydrochloride. Other excipients include Sodium chloride, Sodium hydroxide, Hydrochloric acid, and Water for injections.
Adjuvants have been used with bupivacaine plain to increase its local anaesthetic effect. Alpha-2 agonists such as clonidine or dexmedetomidine can be combined with the local anaesthesia to increase the duration of anesthesia.
Dexamethasone, increases the duration of action when mixed with bupivacaine plain, for nerve blocks, though the mechanism is not well understood.
Mechanism of Action
Bupivacaine causes a reversible blockade of impulse propagation along nerve fibres by preventing the inward movement of sodium ions through the nerve membrane. Amide-type local anaesthetic agents act within the sodium channels of the nerve membrane.
It has rapid onset of action but medium to long duration depending on the dose. It is approximately four times more potent and toxic that lignocaine. The duration of analgesia from the isobaric solution is 3-4 hours in the lower thoracic and lumbar segments.
Bupivacaine plain also produces muscle relaxation in the lower limbs lasting for 3-4 hours, somewhat shorter than the duration of sensory blockade.
During spinal anaesthesia, there may be all in blood pressure and/or intercostal paralysis. This may be due to use of excessive doses, improper positioning of the patient or due to sympathetic blockade.
Pharmacokinetic Properties
Bupivacaine has a pKa of 8.1, and is extensively bound to the plasma proteins (95%). It has high lipid solubility with an oil/water partition coefficient of 27.5. These properties explain its long duration of action.
The maximum plasma concentration is approximately 0.4 mg/L for every 100 mg injected, due to slow absorption from the subarachnoid space and the small dose needed for spinal anaesthesia. Even the maximum recommended dose (20 mg) result in plasma levels of less than 0.1 mg/L.
The total plasma clearance is 0.58 L/min, a volume of distribution at steady state is 73 L, and elimination half-life is 2.7 hours. Liver metabolism is the major form of clearance, and only 6% of bupivacaine is excreted.
Clinical Indication
- Bupivacaine plain is used in intrathecal (subarachnoid, spinal) anaesthesia for surgical and obstetrical procedures
- For lower limb, including hip surgery, that could last for 2 to 4 hours where muscle relaxation is desired.
Dose and Administration:
Bupivaine is administered through local infiltration (post-surgical analgesia), peripheral nerve blocks (dental or other minor surgical procedures, orthopedic surgery), spinal anesthesia (injected into the CSF to produce anesthesia for orthopedic surgery, abdominal surgery, or cesarean delivery), epidural anesthesia for labor pain, and caudal block (anesthesia and analgesia below the umbilicus, mostly for pediatric surgery)
Bupivacaine plain can be used in children. The key difference between the children and adult groups is the relatively high CSF volume in infants and neonates, which means a larger dose/kg, is needed in children to produce the same effect as in adult.
The dosing for bupivacaine for children:
- < 5 kg: 0.40-0.50 mg/kg (dosage)
- 5 to 15 kg: 0.30-0.40 mg/kg (dosage)
- 15 to 40 kg: 0.25-0.30 mg/kg (dosage)
Contraindications
- Allergy or hypersensitivity to amide type local anaesthetics
- Uncorrected hypotension, cardiogenic or hypovolaemic shock
- Spinal stenosis and active disease (e.g. spondylitis, tuberculosis, tumour) or recent trauma (e.g. facture) in the vertebral column.
- Acute active diseases of the cerebrospinal system including meningitis, tumours (primary or secondary), poliomyelitis, subacute combined degeneration of the spinal cord, cranial haemorrhage, demyelinating disease and raised intracranial pressure.
- Inflammation and/or sepsis in the region of the proposed injection or in the presence of septicaemia.
- Coagulation disorders or when taking anti-coagulation treatment.
- Pernicious anaemia with subacute combined degeneration of the spinal cord.
- Due to difficulty in performing spinal anaesthesia in patients with arthritis and other diseases of the vertebral column, bupivacaine may not be used.
Drug Interactions
Certain anti-arrhythmics such as lignocaine, mexiletine, tocainide, are structurally related to bupivacaine and may cause additive systemic toxic effects. Caution is necessary when using them together.
Pregnancy / Lactation
There is no report of any reproductive disturbances or malformations with the use of bupivacaine in pregnancy. However, the dose should be reduced in the late stages of pregnancy.
Bupivacaine enters breast milk in such small quantities that will not cause any risk to the baby.
Adverse Effects of Bupivacaine
Common adverse effects include cardiac disorders (hypotension, bradycardia), gastrointestinal disorders (nausea and vomiting), nervous system disorders (postdural puncture headache), urinary disorders (urinary retention, urinary incontinence).
Less common adverse reactions include nervous system disorders (paraesthesia, paresis, dysaesthesia), musculoskeletal, connective tissue and bone disorders (muscle weakness, back pain).
In rare occasions, especially in extensive (total) spinal blockade, patient may witness crdiac disorders (cardiac arrest), immune system disorders (allergic reactions, anaphylactic shock), nervous system disorders (total spinal block (unintentional), paraplegia, paralysis, neuropathy, arachnoiditis, and respiratory disorders (respiratory depression)
Precautions
- Bupivacaine plain should be stored at 25°C or below. Do not freeze
- Spinal injections should only be made after the subarachnoid space has been clearly identified by lumbar puncture. The injection is given after the clear cerebrospinal fluid (CSF) is seen to escape from the spinal needle or it is detected by aspiration.
- It must be protected from light
- The solution is for single use, and any unused portion should be discarded
- Local anaesthetic agents may react with certain metals such as metal bowls, cannulae and syringes with metal parts after long exposure. Long contact with such metals should be prevented.
References:
- https://www.ncbi.nlm.nih.gov/books/NBK532883/#:~:text=Bupivacaine%20is%20offered%20in%20three%20different%20concentrations:%200.25%25%2C%200.5%25%2C%20and%200.75%25.
- https://www.medsafe.govt.nz/profs/datasheet/m/MarcainSpinalinj.pdf
- https://www.septodontusa.com/product/pain-management-marcaine/#:~:text=Safety%20information%20for%20Marcaine%20includes:%20%20Contraindicated,%20Gluten%20*%20Peanut%2C%20soy%2C%20or%20nut
- https://reference.medscape.com/drug/marcaine-sensorcaine-bupivacaine-343360