The use of inhalational anaesthetic agents date back to 1840s, with the emergence of nitrous oxide (1844), ether (1846), and chloroform (1847). However, these agents were found to be effective but highly flammable or toxic to biological tissues, leading to research and discovery of other inhalational agents like fluorinated ethers and hydrocarbons.
Halothane, discovered in 1951 and was first used clinically in 1956, was found to have favorable safety profile.
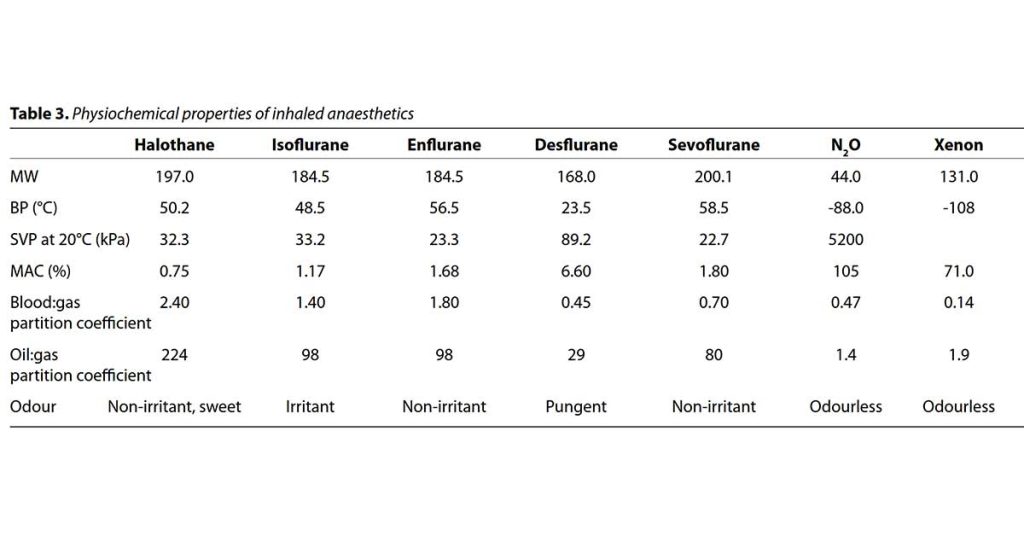
The modern agents used today include the fluorinated ethers isoflurane, sevoflurane, and desflurane and the gas nitrous oxide (N2O). Also, xenon, a noble gas, has a great anaesthetic properties, but the production cost is a huge challenge.
Halothane is still used in mostly low-income countries, but has been replaced with fluorinated ethers: isoflurane, sevoflurane, and desflurane, due to less adverse effect.
Ideal Physical Properties of Inhalational Anaesthetic Agents
- It must not be flammable
- Must be stable without need for preservatives
- Must have high saturated vapour pressure to allow easy vaporization
- Should not interact with anaesthetic equipment (rubber, plastic, glass, metals, soda lime)
- Non-irritant to the respiratory tract and non-pungent to allow gaseous induction
- Easily scavenged
Ideal Pharmacology Properties of Inhalational Agents
- Volatile inhalational anaesthetic agents undergo minimal to no metabolism and are excreted unchanged by lungs. It is not affected by hepatic or renal impairment/disease.
- High oil:gas coefficient to ensure high potency
- Low blood:gas coefficient to facilitate rapid onset of anaesthesia
- Does not interfere with other anaesthetic agents/drugs
- No adverse effects on other organ systems
- Does not trigger malignant hyperthermia
- No toxic effects like allergic reactions, carcinogenicity, and teratogenicity.
Mechanism of Action
Inhalational anaesthetic agents have been used for 170 years, but the mechanism of action is yet unknown. They belong to diverse group with no specific chemical class. Macroscopically, volatile anaesthetic agents prevent both the transmission of noxious stimuli ascending in the spinothalamic tracts to the thalamus and subsequently the cortex, in addition to a neuronal response to pain at a spinal level.
Hypnosis and amnesia most likely result from direct supraspinal depression, especially in the thalamus and reticular formations.
There are two major theories of the mechanism of actions of inhalational anaesthetic agents at molecular level.
- Lipid theory or Meyere-Overton correlation
- Protein theory
Lipid theory or Meyere-Overton correlation
In the early 1900s, Meyer and Overton noticed a linear relationship between log minimum alveolar concentration (MAC) and and lipid solubility. The more soluble a volatile anaesthetic (represented by a higher log oil:gas partition coefficient), the more potent the agent is (lower log MAC).
This led to the theory that volatile anaesthetic agents caused interruption of the usual function of the cell membrane lipid bilayer by penetration and disruption of the molecular arrangement of phospholipids and ion channels.
The more soluble an agent, the more disruption to the membrane at lower doses.
Key Note:
Oil:Gas Partition Coefficient and Minimum Alveolar Concentration relate to potency, while Blood:Gas partition Coefficient relates to speed of onset and offset.
1. Minimum Alveolar Concentration (MAC): The MAC of an inhalational anaesthetic agent is a measure of potency of an inhalational anaesthetic agent and is effectively its ED50 (the administered dose that produces an effect in 50% of the population). MAC is affected by other drugs, patient factors and concurrent illness.
2. Blood:gas partition coefficient: The blood:gas partition coefficient is a measure of solubility in blood, defined as the ratio of anesthetic concentration in blood to that in gas at equilibrium.
Obese patients will experience slower onset of, and emergence from, anaesthesia. This is due to slow, prolonged release of agent from the creation of a larger depot in fatty tissue. The use of less soluble agents with lower blood:gas partition coefficients will be beneficial in such obese patients.
Blood:gas partition coefficients are increased by serum constituents such as albumin, cholesterol, globulin and triglycerides. Infants and older patients with lower levels of these constituents will experience more rapid onset of anaesthesia.
3. Oil:gas coefficient: This is a a measure of the solubility of the inhalational anaesthetic agent in fat/lipids compared to gas at equilibrium. A higher coefficient signifies higher lipid solubility, which directly correlates to greater anesthetic potency (lower MAC). It determines how much an agent distributes into fatty tissues.
Protein theory
This theory replaces the popular lipid theory. Anaesthetic agents act by inhibiting excitatory (serotonergic, neuronal nicotinic and N-methyl-D-aspartate (NMDA)) and activating inhibitory (g- aminobutyric acid (GABA)A and glycine) ion channels which are distributed throughout the central nervous system (CNS).
This results in the pre-synaptic inhibition of neurotransmitters and/or a reduction post-synaptic response thresholds.
Volatile anaesthetic agents are thought to activate the GABAA ion channel by binding to the alpha subunits causing a conformational change resulting in increased chloride conductance and thus hyperpolarization of the cell membrane.
Potassium channels that set resting membrane potentials of pre and post-synaptic membranes in the CNS are believed to be site of action of volatile agents.
Examples of Modern Anaesthetic Agents with their Properties
The addition of a fluorine molecule to carbon decreased flammability of earlier inhalational anaesthetic agents. The fluorine–carbon bond was more stable than the carbon–carbon bond, and seemed to undergo less biological metabolism, and was therefore associated with less organ toxicity.
The fluorinated anaethetic compound first marketed was fluroxene. It was introduced in 1954 and used until 1975. It has favorable effects such as low solubility in blood and minimal tendency
to depress cardiovascular function, however, it can cause nausea and vomiting, and at high concentration, it was flammable.
In today’s practice, fluorinated compounds are the inhalational anaesthetic agents of choice. They include:
- Halogenated hydrocarbon like Halothane,
- Halogenated ethyl methyl ethers like Enflurane, Isoflurane,and Desflurane,
- Polyfluorinated isopropyl methyl ether: Sevoflurane
Isoflurane and enflurane are structural isomers of each other. The inorganic gas, nitrous oxide is not fluorinated nor available in DEPMEDS-equipped hospitals, but is included in the list. Also, the elemental gas (Xenon) is included.
Halothane
Halothane is non-irritant, and has sweet odor, and as such can be used for gaseous induction. It in unstable when exposed to light, corrode certain metals, and also dissolves into rubber. Halothane is prepared with 0.01% thymol to prevent liberation of free bromine and decomposition on exposure to light.
Halothane was made in 1951 and first used in the UK in 1956. It is the first ‘modern’ volatile agents. Halothane is used for both induction and maintenance of general anaesthesia.
Effect
Respiratory: Minute ventilation is depressed largely due to decreased tidal volume and normal response to hypoxia and hypercarbia are blunted. Since halothane leads to bronchodilation, it can be used in asthmatic patients.
Cardiovascular: Bradycardia is produced by increased vagal tone, depressed sino atrial and atrio-ventricular activity. The myocardium is directly depressed and systemic vascular resistance (SVR) is reduced. Halothane sensitizes the heart to catecholamines. This may lead to arrhythmias and the quantity of adrenaline used for infiltration should be limited.
Central nervous system: There is increase in cerebral blood flow more than any other volatile agent, which increases intra-cranial pressure (ICP).
Metabolism
Around 25% of the halothane undergoes oxidative metabolism by hepatic cytochrome p450 to produce trifluoroacetic acid, Br- and Cl -. However, when the liver is hypoxic, reductive metabolism predominates producing F-.
Toxic Effect
Halothane may lead to hepatic damage. This damage is of two forms – the reversible form and the fulminant hepatic necrosis (‘halothane hepatitis’).
If halothane has been given three months prior, it should be avoided. Also, if the patient has adverse reaction to it, or in cases of pre-existing liver disease.
Isoflurane
This inhalational anaesthetic agent is used widely to maintain anaesthesia and to treat severe bronchospasm. Isoflurane has a pungent smell and can lead to upper airway irritability and breath-holding.
Isoflurane was first synthesized in 1965 and first used clinically in 1980 due to concern about carcinogenicity. It is stored in amber color bottle.
Effect
Respiratory: Isoflurane depresses ventilation more than halothane, but less than enflurane. Minute ventilation is decreased, respiratory rate and PaCO2 are increased. It does not cause bronchodilation. Due to the pungent smell and breath-holding, it is not used for gaseous induction.
Cardiovascular: The major effect is to reduce systemic vascular resistance (SVR) leading to compensatory tachycardia and only a small decrease in myocardial contractility. Isoflurane’s effect on the ATP-dependent potassium channels may have protective effect on the myocardium.
Central nervous system: Isoflurane produces the best balance in reduced cerebral oxygen requirement and minimal increase in cerebral blood flow when compared to other volatile agents.
Toxic Effect
Carbon monoxide may be produced by a reaction between the -CHF2 group and dry soda lime (or baralyme).
Enflurane
Due to the discovery of newer inhalational anaesthetic agents with better profile, the use of enflurane has decreased. Only 2% is metabolized by hepatic cytochrome P450 producing F- ions. However, the level of F- ions produced are low and rarely cause reversible nephropathy.
Effects
Respiratory: Enflurane causes more depression of ventilation than the other agents, with a reduction in minute volume and increase in PaCO2. Response to hypercarbia is blunted.
Cardiovascular: There is increase in heart rate but fall in cardiac output, contractility, systemic vascular resistance and blood pressure.
Central nervous system: Enflurane is avoided in epileptic patients as high concentrations of enflurane in the presence of hypocarbia produce 3Hz spike and wave pattern on the EEG consistent with grand mal activity.
Toxicity
May cause hepatic damage. It may be avoided in patient with renal impairment.
Desflurane
This agent is used for the maintenance of general anaesthesia. It has ethereal and pungent odour.
Desflurane has a relatively low boiling point (23.5°C), making it very volatile. This temperature is close to ambient temperature in many theatre settings, full vapour saturation cannot be achieved with a conventional vaporizer. A Tec 6 vaporizer is used to heat the inhalational agent to 39°C at a pressure of 2 atmospheres, to ensure full vapour saturation.
Desflurane is ideal for long procedures where rapid wake-up is important (after neurosurgery) due to its ow blood:gas partition coefficient property. Desflurane was first used in humans in 1988, and introduced into clinical practice in 1993.
Effects
Respiratory: It has a potent odour, causes cough and breath-holding, hence not suitable for induction. Desflurane has similar respiratory effects like other agents causing a rise in PaCO2 and a fall in minute ventilation (the effect is more with halothane, and less with isoflurane and enflurane).
Cardiovascular: Has similar effect as isoflurane. However, desflurane may produce tachycardia and hypertension at concentration above 1 MAC.
Vascular resistance falls in the cerebral and coronary circulations, therefore, care should be taken with ischaemic heart disease patients.
Sevoflurane
With the exception of halothane, sevoflurane undergo hepatic metabolism by cytochrome p450 (2E1) more than any other inhalational anaesthetic agent. Sevoflurane is used for maintenance and induction of anaesthesia, and to treat severe bronchospasm.
It has pleasant smell. Sevoflurane is stored in amber coloured polyethylene napthalate bottles with added concentration of 300 ppm of water to prevent attack by Lewis acids and release of toxic hydrofluoric acid.
First synthesized in 1968, clinical use started in 1990 due to initial concerns over issues with biotransformation and stability with soda lime.
Effects
Respiratory: Sevoflurane has a pleasant odour and a relatively low blood:gas partition coefficient, making it useful for induction. There is depression of ventilation in a predictable manner with a rise in PaCO2 and a fall in minute ventilation.
Cardiovascular: There is no change in heart rate and contractility, but a fall in SVR leads to lowering of blood pressure. Also, there is reduction in vascular resistance in the cerebral and coronary circulations.
Central nervous system: Evidence suggests that children children exhibit a higher incidence
of post operative agitation and delirium compared to halothane.
Toxic Effects
Administering sevoflurane in a closed circle system using soda lime or baralyme leads to the production of compounds A-E. Only compounds A and B are present in significant quantities. However, the levels of these compounds are not associated with renal impairment.
Ether (Diethyl ether)
Ether is a rarely used and inexpensive agent made from sugar cane (ethanol). It is the volatile agent of choice when general anaesthesia is needed but oxygen is not available. It is used in certain countries.
Ether also has analgesic effect.
Effects
Respiratory: It leads to stimulation of respiration. Higher concentration of ether depresses the respiration before the heart. Ether causes bronchodilation, and can be used as a sole anaesthetic agent producing good abdominal muscle relaxation.
Antisialogogue premedication is needed as ether stimulate salivation.
Ether has slow onset and recovery. It also slows down induction of anaesthesia as the vapour is unpleasant to breathe initially and irritates the bronchial tree.
Cardiovascular: Increase in cardiac output and blood pressure due to its sympathomimetic effect mediated by adrenaline release.
Other effects: Ether can induce nausea and vomiting more than other agents. Using endotracheal
tube during relaxant anaesthesia reduces the nausea and vomiting effect.
Since ether causes little uterine relaxation, it is useful during caesarean section. The baby tolerates it, and the uterine contracts well. However, it is avoided in moderate or severe pre-eclampsia due to its sympathomimetic activity.
Caution
Ether is inflammable in air and explodes with mixed with oxygen. A flame or an electrical spark produced by diathermy or static electricity can ignite it. Ether vapour is inflammable within the patient (lungs, airway or stomach full of vapour) or outside the patient within 25cm of the anaesthetic circuit.
Scavenging is done to avoid contact between heavy inflammable ether vapour and diathermy apparatus or other electrical devices that may spark.
Nitrous Oxide (N2O)
Nitrous oxide, an odorless inorganic gas, known as ‘laughing gas”, has a a high minimum alveolar concentration, MAC. It is used in combination with other inhalational agents, or with O2 as entonox. However, nitrous oxide interferes with DNA synthesis even with little exposure. N2O is stored as a liquid in French blue cylinders.
The filling ratio of the cylinders is 0.75 in temperate regions, but reduced to 0.67 in tropical areas to
prevent cylinder explosions.
Mechanism of action: NMDA-receptor antagonist, with little or no effect at GABAA receptors. It also stimulates the dopamine, a1– and a2-adrenergic and opioid receptors
Effect:
Respiratory: Leads to small fall in tidal volume which is offset by an increased respiratory rate.
Cardiovascular: There is mild direct myocardial depression which is offset by an increase in sympathetic activity via its central effects.
Central nervous system: Cerebral blood flow (CBF) is increased.
Toxic Effect:
N2O oxidises the cobalt ion in vitamin B12 preventing its action as a cofactor for methionine synthase. This reduces the synthesis of methionine, thymidine, tetrahydrofolate and DNA. Exposure to nitrous oxide leads to megaloblastic changes in bone marrow. Exposure for a long time may result in agranulocytosis and neurological syndromes.
Nitrous oxide is mostly avoided in the first trimester.
Xenon (Xe)
Xenon (Xe) is an inert, odourless gas with a high MAC and very low blood:gas partition coefficient, which result in faster onset and offset of action than desflurane or N2O. Xenon also has an analgesic effect, neuroprotective properties, causes little or no cardiovascular depression and is not metabolised.
Xenon, like N2O and ketamine, acts by non-competitive inhibition of NMDA receptors in the CNS. N2O and xenon has little or no effect on GABAA receptors.
The high cost of production and the rarity of xenon means it is mostly used as a research agent and not for clinical use.
References
- https://resources.wfsahq.org/wp-content/uploads/uia-24-2-Pharmacology-of-Inhalational-Anaesthetics.pdf
- https://www.ncbi.nlm.nih.gov/books/NBK554540/
- https://anaesthetics.ukzn.ac.za/wp-content/uploads/2024/04/Volatiles-AICM-2019.pdf
- https://www.frcanotes.com/Partition_coefficients.html
- https://www.anaestheasier.com/inhalational-anaesthetic-agents/