The World Health Organisation Guideline Development Group of international experts has recommended approving two new covid drugs in the BMJ. This is after a trial that involved over 4000 patients with a different level of severity of Covid-19 infections.
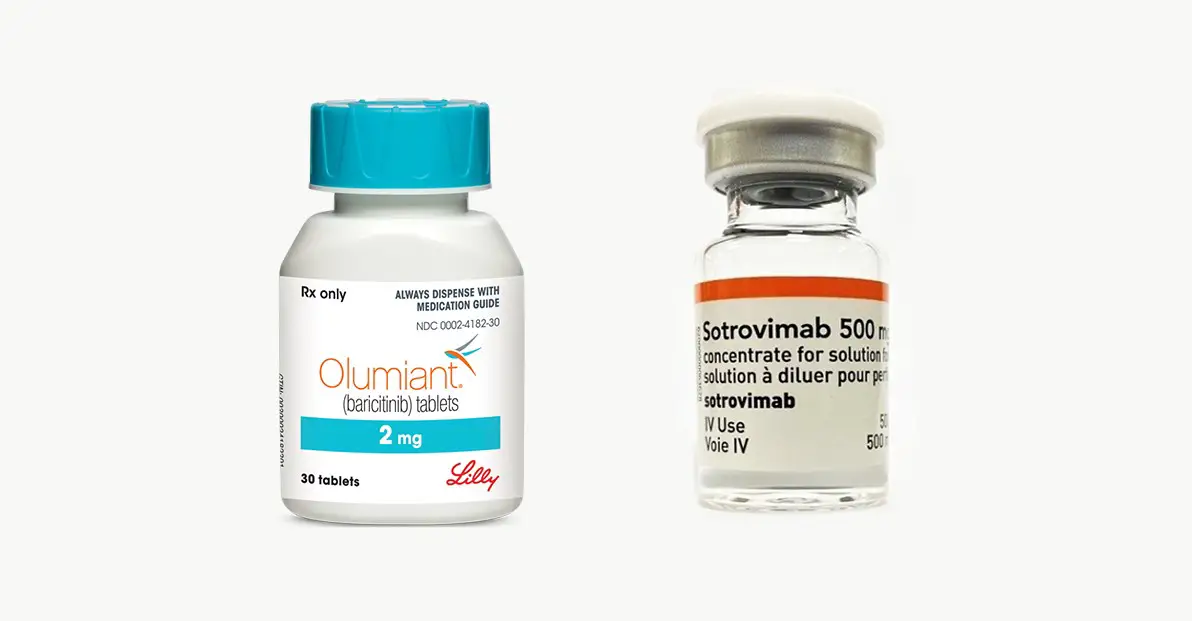
The drug, Baricitinib, a Janus kinase (JAK) inhibitor, is used to treat rheumatoid arthritis. WHO recommends the use of the drug in combination with systemic corticosteroids in severe and critical cases of Covid-19 disease. This is because of observed evidence that it improves survival and reduces the need to use a ventilator.
Baricitinib has the same effect just like other arthritis drugs, the interlukin-6 inhibitors. They can use any of the options if available or less costly based on the recommendation. However, both of them cannot be used together.
The WHO experts also warned that other JAK inhibitors, such as ruxolitinib, and tofacitinib, should not be administered because they do not offer any benefit to patients, but result in adverse effects.
READ ALSO: A first ever spinal cord stem cell transplant conducted by the Japanese team
For non-severe cases of Covid-19, they made a recommendation for the use of monoclonal antibody sotrovimab to reduce the risk of hospitalisation. They also made a conditional recommendation for Casirivimab-imdevimab, another monoclonal antibody for non-severe cases of covid-19
Using one monoclonal antibody over another has no added benefit, as there is no sufficient data to justify its effectiveness. Also, the antibody might not be effective on a new variant of Covid, such as omicron.
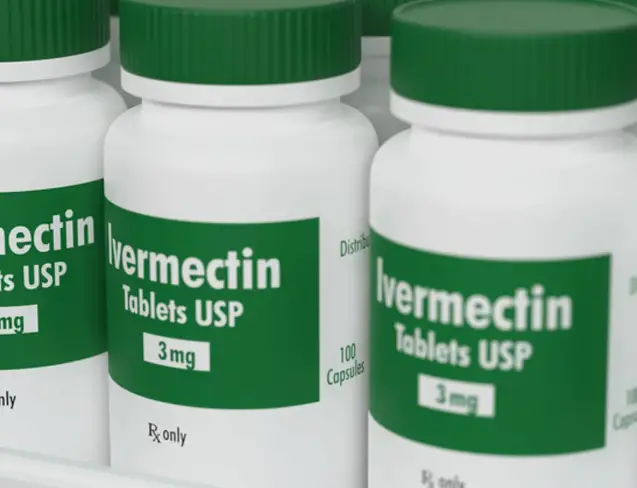
The WHO has previously warned against the use of convalescent plasma, ivermectin and hydroxychloroquine in treating covid-19 patients at any stage of the disease.
Source: Medicalxpress