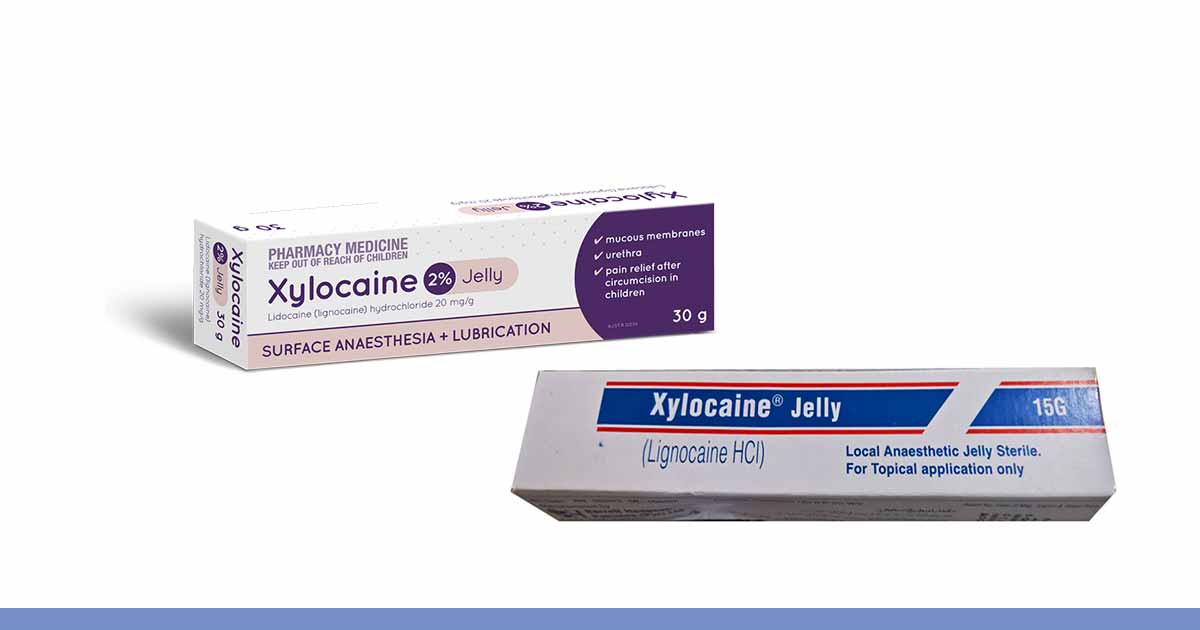
Xylocaine jelly, or lidocaine hydrochloride jelly USP, lignocaine jelly is a topical anaesthetic agent that contains lidocaine. It is used for surface anesthesia and lubrication during medical procedures such as proctoscopy and rectoscopy, tracheal intubation, in the nasal and pharyngeal cavities during endoscopic procedures such as gastroscopy and bronchoscopy.
It is also used in both male and female urethra during cystoscopy, catheterization, exploration by sound and other endourethral operations. Another use is in treatment of pain in cystitis and urethritis.
Lidocaine jelly or K-Y jelly can either be used to lubricate tracheal tubes during tracheal intubation for general anesthesia,
Lidocaine provide local anesthetic effect by stabilizing the neuronal membrane. It achieves this by inhibiting the ionic fluxes required for the initiation and conduction of the impulse. This amide type of anesthetic act within the sodium channels of the nerve membrane.
Lidocaine action is within 3 to 5 minutes, and effect last for 20 to 30 minutes. However, it is ineffective in intact skin. Due to the depressant effect, it may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure.
Lidocaine has a total plasma clearance of 0.95 L/min and a volume of distribution at steady state of 91 L.
It is metabolized by the liver, and the primary metabolite in the urine is the conjugate of 4-hydroxy-2, 6-dimethylaniline. Lidocaine crosses the blood-brain barrier and the placenta.
What is in Xylocaine Jelly?
Xylocaine jelly 2% (lidocaine hydrochloride) is a clear and slightly colored jelly. It comes in aluminum tube.
The active ingredient is lidocaine hydrochloride (20 mg/mL) while the vehicle is water thickened with hydroxypropyl methylcellulose (hypromellose).
There is also sodium hydroxide and/or hydrochloric acid to adjust pH to 6.0-7.0.
The preservatives are methylparaben and propylparaben. The water-soluble base has high viscosity and low surface tension. The mixture of lidocaine and hypromellose helps to maximize contact with the skin mucosa, and lubricate for instrumentation.
Uses of Lidocaine Jelly
- Anesthetic lubrication for endotracheal Intubation, (both oral and nasal): Apply a moderate amount of the jelly at the external surface of the endotracheal tube (not inside the lumen of the tube) shortly before use.
- Limit pain during urological procedures in the male (20 mL) and female (5-10 mL of jelly) adult urethra
- Nasal and pharyngeal cavities in endoscopic procedures such as gastroscopy and
bronchoscopy. 10-20 mL, but not exceeding 400 mg total doses. - Topical treatment of painful urethritis and cystitis
- Proctoscopy and rectoscopy up to 20 mL, but not exceeding 400 mg total doses.
Dosage of Xylocaine Jelly
Before using xylocaine jelly, consider the age, weight, and the physical condition of the person. Exercise caution in using lidocaine in children less than two years, elderly, debilitated persons, as there are fewer data about their safety in this age bracket.
The dosage of lidocaine depends on the area to be anesthetized, the blood flow and supply of blood vessels in the area, tolerance, and the technique employed.
The lowest effective dose should be considered
Maximum dosage of Lidocaine
In a 12-hour interval, do not give more than 600 mg of lidocaine or give 4 doses within 24 hours. 400 mg (20 mL) is a safe dose for oral use, while 800 mg (40 mL) is safe for the bladder and urethra.
Though it is difficult to calculate the maximum dosage for children, weight, and age should be considered. For children less than ten years, use the standard pediatric drug formulas (e.g., Clark’s rule). For instance, a child weighing 50 lbs will not exceed 75 to 100 mg by Clark’s rule.
In children under 12 years, lidocaine should not exceed 6 mg/kg of body weight or 3 mL per 10 kg weight or more than 4 doses within 24 hours.
Median Lethal Dose (LD50) of Lidocaine:
In non-fasted female rats, the LD50 of lidocaine salt was 459 (346 to 773) mg/kg, while it was 214 (159 to 324) mg/kg in fasted female rats.
Drug Interaction with Lidocaine
Patients who take lidocaine concurrently with these listed medications may develop methemoglobinemia, and other drug reactions
- Anticonvulsants like phenytoin, phenobarbital, sodium valproate
- Nitrates/Nitrites like nitric oxide, nitroglycerin, nitroprusside, nitrous oxide.
- Antimalarials such as chloroquine, primaquine
- Local anesthetics like articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine
- Antibiotics like dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides
- Other medications like acetaminophen, metoclopramide, quinine, sulfasalazine
- Class I antiarrhythmic drugs such as mexiletine due to additive effect
- Class III antiarrhythmic drugs, e.g., amiodarone
- Strong Inhibitors of CYP1A2 and CYP3A4 like fluvoxamine, erythromycin and Itraconazole, reducing the clearance.
- β-blockers and cimetidine, reducing the clearance.
Contraindication
Do not use lidocaine jelly in individuals with hypersensitivity to amide-type local anaesthetic, or other ingredients in the jelly.
Also, people with known hypersensitivity to the preservatives in the formulation – methylparaben and/or propylparaben, or their metabolite, para amino benzoic acid (PABA). Parabens are also avoided in people with allergy to ester local anesthetics, as it is metabolised to PABA.
Caution
Use lidocaine with caution in patients with sepsis or severely traumatized mucosa at the application site, as there could be rapid absorption.
Do not lubricate the endotracheal stylettes as the jelly may be introduced into the inner lumen. It will dry and leave residues which clump and narrow the lumen or cause occlusion.
Lidocaine may cause methemoglobinemia with symptoms such as paleness, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue. The risk of methemoglobinemia is increased in patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents.
If methemoglobinemia occurs, give oxygen therapy, hydration. In severe conditions, give methylene blue, exchange transfusion, or hyperbaric oxygen.
Use lidocaine with caution in patients with epilepsy, impaired cardiac conduction, bradycardia, impaired hepatic or renal function and in severe shock.
Pregnancy, Delivery, and Labour
Lidocaine is excreted in human milk. Take caution while giving to nursing mothers and also in pregnancy, labor.
Children
Use with caution in children. Consider the age, weight, and physical condition. In children less than 2 years, take caution, as there are fewer data to guarantee safety.
Adverse Effects of Lidocaine Jelly
Lidocaine, like other amide local anesthetic agents, may cause dose related effect like excessive plasma level from high dosage, or hypersensitivity, diminished tolerance from patient.
Serious adverse effects are systemic in nature. Rarely, endotracheal tube occlusion may occur when there are dried particles of the jelly in the inner lumen of the tube.
Allergic reactions such as edema, cutaneous lesions, urticaria, and anaphylactic reaction may occur from sensitivity to the lidocaine or other ingredients in the formulation.
There are central nervous system effects which may be excitatory or depressant in nature. They are light headiness, vomiting, confusion, drowsiness, nervousness, apprehension, euphoria, dizziness, tinnitus, blurred or double vision, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression, and arrest.
There may be depression of the cardiovascular system causing bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
Managing Lidocaine Overdose
Excess levels of lidocaine may result in emergency situations. The first step in management is to monitor cardiovascular, respiratory vital signs, and level of consciousness. Administer oxygen when any change occurs.
Manage convulsion first by maintaining airways and ventilation. If the convulsion persists, a small increments of intravenous ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) should be administered.
Supportive treatment for the circulatory depression may involve intravenous fluids and a vasopressor like epinephrine (0.1-0.2 mg as intravenous or intracardial injections)
Convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias, and cardiac arrest when left untreated. If cardiac arrest occurs, standard cardiopulmonary resuscitation measures should be used.
References:
- https://dailymed.nlm.nih.gov/dailymed/getFile.cfm?setid=317a8784-5fdd-429f-9b74-14d89f372a32&type=pdf
- https://pdf.hres.ca/dpd_pm/00005549.PDF